 The ideal bond angles in a trigonal pyramid are based on the tetrahedral electron pair geometry. You might need the complete molecular shape (molecular geometry) table.\)) with the nitrogen atom at the apex and the three hydrogen atoms forming the base. 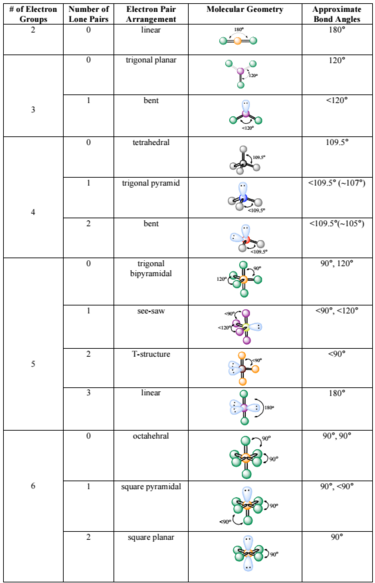
The hybridization of phosphorous in PCl 3 is sp 3. In the PCl 3 Lewis dot structure, a total of 10 lone pairs and 3 bond pairs are present. VSEPR theory predicts the three-dimensional arrangement of atoms in a molecule. Key Points Linear: a simple triatomic molecule of the type AX2 AX 2 its two bonding orbitals are 180° apart. The electron geometry of PCl 3 is Tetrahedral, as its central atom, is surrounded by the 4 regions of electron density. PRACTICE PROBLEMS : Use the electron dot structure (Lewis structure) to determine the molecular shape (molecular geometry). The molecular geometry or shape of PCl 3 is a Trigonal pyramid. Molecular Shape of BrH 3 (Answer): T-SHAPED Fluorine belongs to the family of halogen in group 17 and has a valency of 7. click for print view14. Electron and molecular geometry on central atom. Oxygen belongs to group 16, the chalcogen family, and has a valency of 6. Geometry molecular electron vsepr molecule theory sampletemplates lone vs atoms predict xef2 bonded socratic steric trigonal electrons Geometry molecular electron chart bonding. The first and foremost step is to calculate the total number of valence electrons in an OF2 molecule. Step 1: Find the Total number of Valence Electrons. Lone pair electrons of central atom: 2 pairs (4 electrons total)īond Angle of BrH 3 (Answer): Less than 90 degrees Steps to form OF2 Lewis Structure Diagram. The number of valence electrons in the central atom must be determined. Molecular Shape of AsCl 3H 2 (Answer): TRIGONAL BIPYRIMIDAL It should be the atom with the highest electronegative potential. Molecular Shape of PH 5 (Answer): SQUARE PYRAMIDALīond Angle of AsBr 3H 2 (Answer): 90 and 120 degrees Lone pair electrons of central atom: 1 pair (2 electrons total) Molecular Shape of SF 6 (Answer): OCTEHEDRAL Electron Geometry: Describes the arrangement of bonds and lone pairs around a central atom. Lone pair electrons of central atom: 0 pairs VIDEO Molecular Shape from Lewis S tructure Examples 2. Use the complete molecular shape (molecular geometry) table below to answer the examples and practice problems.Įxamples: Use the complete molecular shape (molecular geometry) table along with the electron dot structure (Lewis structure) to determine the molecular shape (molecular geometry). You identify these molecular shapes and bond angles from their electron dot structures by counting both the atoms bonded to the central atom and the lone pair electrons of the central atom. They include shapes like triginal bipyrimidal and octahedral but only make sense if you have explored the electron dot structures (Lewis structures) beyond the octet of 8. VESPR stands for valence shell electron pair repulsion. It applies a theory called VESPR for short. Those molecular shapes and bond angle beyond Tetrahedral (4 bonds – or electron clouds) are the more advanced shapes. Molecular geometry is a way of describing the shapes of molecules. What are the more advanced molecular shapes (molecular geometry)?īefore taking on this section, make sure you have gone over the completing the octet college section. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
